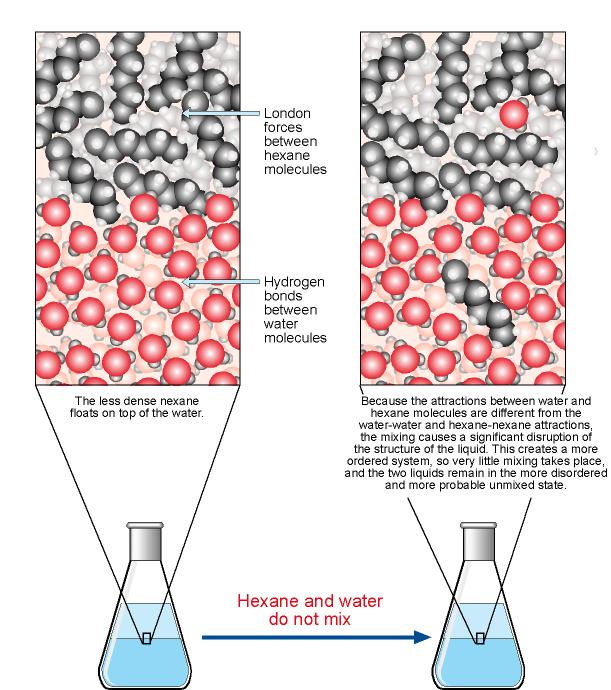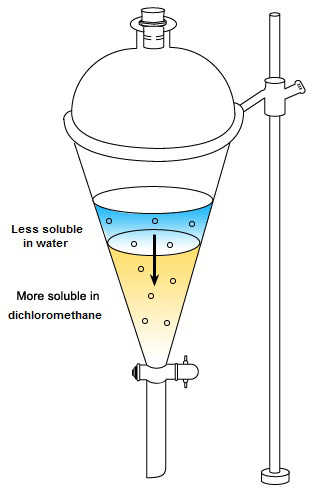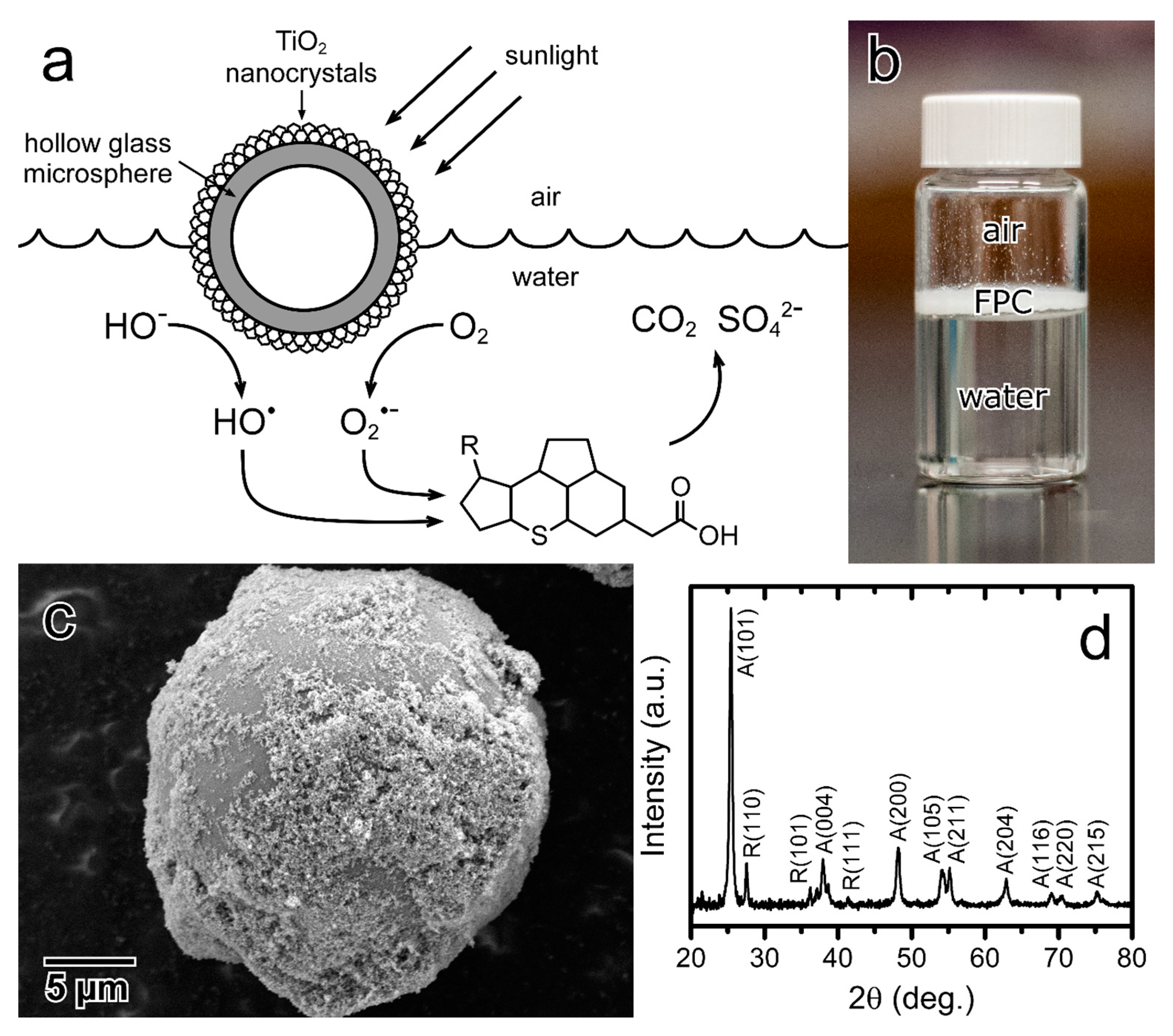
Water | Free Full-Text | Floating Photocatalysts for Passive Solar Degradation of Naphthenic Acids in Oil Sands Process-Affected Water

Dual Superlyophobic Materials for Under-Liquid Microfluid Manipulation, Immiscible Solvent Separation, and CO2 Blockage | ACS Applied Materials & Interfaces

China Main Uses Of Dichloromethane DCM CAS 75-09-2 Manufacturers, Factory - Wholesale Discount - QIBO

Donor–Acceptor‐Type Organic‐Small‐Molecule‐Based Solar‐Energy‐Absorbing Material for Highly Efficient Water Evaporation and Thermoelectric Power Generation - Cui - 2021 - Advanced Functional Materials - Wiley Online Library

China Main Uses Of Dichloromethane DCM CAS 75-09-2 Manufacturers, Factory - Wholesale Discount - QIBO

Methylene Chloride with Hscode 290312000 for Paint Thinner - China Good Quality, Liquid | Made-in-China.com
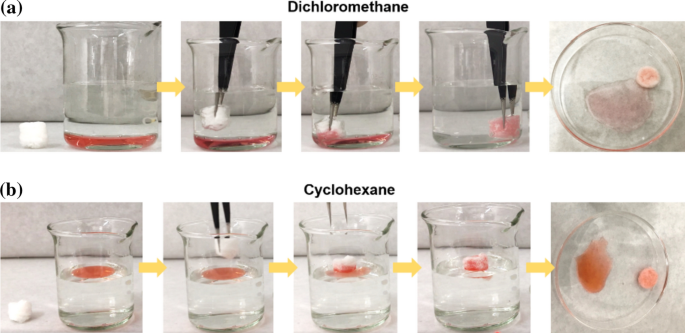
Composite sponges fabricated by silk fibers and alginate for efficient oil/ water separation | SpringerLink